- Abstract
- Overview of pseudopotentials
- Properties of pseudopotentials
- Example in the paper
- Conclusion
- Extended reports
Abstract
Pseudopotentials help researchers to release the calculation workload by reducing the electron number involved in calculation, focusing on the relevant valence electrons, reducing the nodes of wave function, which results in a relatively simple Fourier series when describing the atom. It has a cut-off radius $r_{c}$ and beyond this region, the real and pseudopotentials shows excellent identity and gives good transferability as well, which means the pseudopotentials should resemble the valence obits of various atoms in various chemical environment.
Overview of pseudopotentials
When solving the Schrodinger equation $(T+V)\varphi=\epsilon\varphi$, where the $V$ is the sum of self-consistent core potential and valence potential, the solution would be hard to obtained as the complex potential $V$ has strong wiggles, especially in the core region. So pseudopotential $V_{l}$ is needed to substitute the real potential $V$ with lots of wiggles to solve this Schrodinger equation $(T+V_{l})w_{l}=\epsilon w_{l}$ , where the $w_{l}$ is wavefunction obtained from pseudopotential.
There are several kinds of pseudopotentials, most popular ones are norm-conserving pseudopotentials and ultrasoft pseudopotentials. In the essay, norm-consering is mainly discussed.
For norm-conserving pseudopotentials, the total charge number inside the cutoff region $r < r_{c}$ of modified wavefunction $w_{l}$ should be the same as that of original wavefunction $\varphi$. The pseudopotential generated in this method could be used for wavefuntions in both real and reciprocal space, and make it possible to compare with experimental data. In addition, some approximations were used to simplify calculation like frozen core approximation to individually calculate the charge density of core and valence region and get the sum, $\rho(r)=\rho_{c}(r)+\rho_{v}(r)$.
Properties of pseudopotentials
Four important properties
From the essay, there are four important properties of pseudopotentials that they must follow to obtain the identity between real and pseudopotentials:
-
For a specific atomic configuration, the real and pseudo valence eigenvalues fit with each other.
-
When $r>r_{c}$, real and pseudo atomic functions fit with each other.
-
When $r>r_{c}$, the integrals from $0$ to $r$ of the real and pseudo charge density are the same.
-
When $r>r_{c}$, the logarithmic derivative of the real and pseudo wave function and their first energy derivatives are the same.
Property 1 is quite important when we take the whole periodic table into consideration.
Property 2 ensures the identity so that we could use the pseudopotential solve the Schrodinger equation.
Property 3 and 4 both show the transferability of the pseudopotentials. Property 3 presents that the real and pseudo core have same charge numbers within the cutoff region. For property 4, to verify the goodness of transferability, comparison of logarithmic derivatives of wave functions helps a lot. If the logarithmic results are nearly identical, the pseudopotential has a good transferability.
Derivation
Starting from the Schrodinger equation,
\[\left(-\frac{\hbar^{2}}{2}\nabla^{2}+V(r)\right)\varphi_{i}=\epsilon_{i}\varphi_{i} \tag{1}\label{Schrodinger}\]with density function theory, density $\rho(r)=\sum\varphi_{i}^{*}\varphi_{i}=\sum\lvert\varphi_{i}(r)\rvert^{2}$,
\[V(r)=V_{ext}(r)+e^{2}\int\frac{\rho(r^{'})}{\lvert r-r^{'}\rvert}d^{3}r^{'}+v_{xc}[\rho(x)]\label{DFT}\]the second term is Hartree potential and the third term is exchange-correlation potential.
As mentioned in previous part, the frozen core approximation is used, so $\rho(r)=\rho_{c}(r)+\rho_{v}(r)$. When the core and valence density do not overlap, the term $v_{xc}[\rho(x)]$ approximates $v_{xc}[\rho_{v}(x)]+v_{xc}[\rho_{c}(x)]$.
The orthogonalized-plane-wave(OPW)-like pseudopotential has an important identity
\[2\pi\left[\left(r\varphi\right)^{2}\frac{d}{d\epsilon}\frac{d}{dr}\ln\varphi\right]_{R}=4\pi\int_{0}^{R}\varphi^{2}r^{2}dr \tag{2}\]where $R$ indicates the reference state, $\varphi$ is the solution of Schrodinger equation at energy $\epsilon$. So the atomic scattering property is well preserved.
The Schrodinger-like equation of the valence electrons is,
\[\left\{ -\frac{1}{2m}\frac{d^{2}}{dr^{2}}+\frac{k(k+1)}{2mr^{2}}+V_{k}^{ps}(r)\right\} u_{k}(r)=\epsilon_{k}u_{k}(r)\tag{3}\]while the quantum number $k=l$ for $j=l-\frac{1}{2}$, or $k=-(l+1)$ for $j=l+\frac{1}{2}$. In non-relativistic approximation we could obtain the equation by using $l=k$,
\[\left\{ -\frac{1}{2m}\frac{d^{2}}{dr^{2}}+\frac{l(l+1)}{2mr^{2}}+V_{l}^{ps}(r)\right\} u_{l}(r)=\epsilon_{l}u_{l}(r)\tag{4}\]A first-step pseudopotential $V^{ps}(r)$ is obtained by adding a cutoff at the core
\[V_{1l}^{ps}(r)=\left[1-f\left(r/r_{cl}\right)\right]V(r)+c_{1}f(r/r_{cl}) \tag{5}\]where $r_{cl}$ is the referential core radius for quantum number $l$. The $f(x)$ is the cutoff function which ensure the smoothness of the potential.
The choice of cutoff functions:
\[f(x)=\exp(-x^{\text{\lambda}}) \tag{6}\]That is a numerical solution that will precisely fit the real condition, in this essay $\lambda=4$ and in some others mentioned that $\lambda=3.5$, and both of them show good identity when $r>r_{c}$.
A nodeless solution that would be obtained is $w_{1l}$, solving radial Schrodinger equation with this potential by changing the parameter $c_{1}$ in this equation and the $w_{1l}$ can be normalized with a multiplicative constant $\gamma_{l}$,
\[\gamma_{l}w_{1l}(r)\rightarrow u_{l}(r),\;r>r_{c} \tag{7}\]Then, we need to introduce the norm-conservation concept into the pseudopotential by modifying the wavefunction $w_{1l}(r)$, adding an adjustment within the core region to get $w_{2l}(r)$
\[w_{2l}(r)=\gamma_{l}[w_{1l}(r)+\delta_{l}g_{l}(r/r_{cl})] \tag{8}\]where $\delta_{l}$ is a phase shift generated from the potential and shows how it shifts compared with $w_{1l}$ and it could be obtained by solving the quadratic equation of the normalization of $w_{2l}$,
\[\int_{0}^{\infty}w_{2l}(r)^{2}dr=\gamma_{l}^{2}\int_{0}^{\infty}[w_{1l}(r)+\delta_{l}g_{l}(r/r_{cl})]^{2}dr=1 \tag{9}\]The norm correction $(\gamma_{l}^{2}-1)$ has a relatively small value around $10^{-2}\sim10^{-3}$.
$g_{l}(x)$ here should vanish as fast as $x^{l+1}$ for small $x$, and when $r>r_{c}$, the shape of $\omega_{1l}$ should be preserved since it already fits the real solution. Then $g_{1}(x)$ is chosen to be:
\[g_{l}(x)=x^{l+1}f(x)=x^{l+1}\exp(-x^{4}) \tag{10}\]Thus, the final pseudopotential $V_{2l}^{ps}$ with a nodeless eigenfunction $w_{2l}$ at eigenvalue $\epsilon_{l}$ could be obtained by inverting the Schrodinger equation,
\[V_{2l}^{ps}(r)=V_{1l}^{ps}(r)-\left[g_{l}^{-1}(r)Dg_{l}(r)+V_{1l}^{ps}(r)-\epsilon_{l}\right]/\left\{ 1+w_{1l}(r)/\left[\delta g_{l}(r/r_{cl})\right]\right\} \tag{11}\]where
\[D=-\frac{1}{2m}\frac{d^{2}}{dr^{2}}+\frac{l(l+1)}{2mr^{2}}\]Example in the paper
From the figure, we can see how identical they are when $r>r_{c}$.
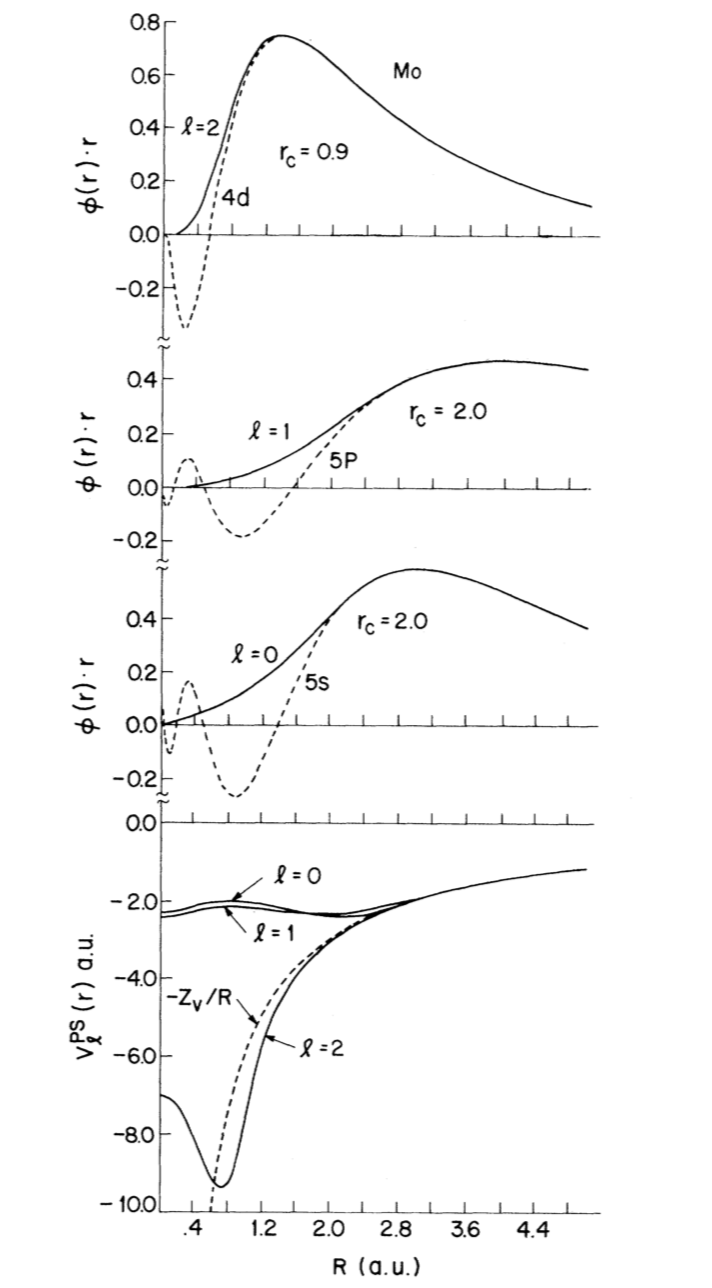
Figure 1. Comparison of pseudo wave functions (solid lines) and ab initio full-core atomic valence wave functions (broken) lines for Mo.
The logarithmic derivatives of wavefunctions shows the transferability by how identical they are.
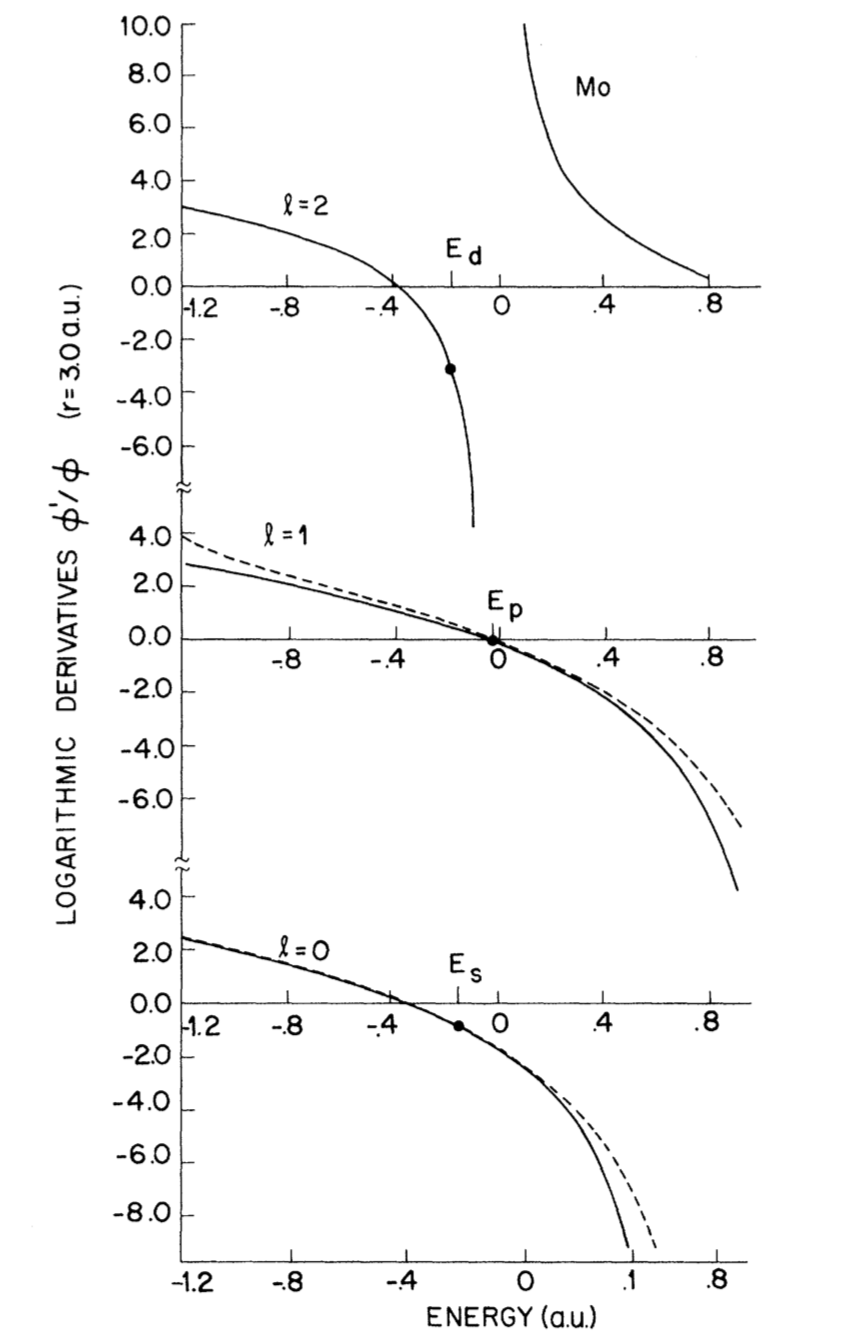
Figure 2. Energy dependence of logarithmic derivatives at $r=3.0 a.u.$ for Mo ab initio full-core atomic wave functions (broken lines) and pseudo wave functions (solid lines).
By comparing Figure 1 and Figure 2, the smaller the cutoff radius, the better identity the logarithmic derivatives will have, and the better transferability will be obtained.
Conclusion
Pesudopotential is a useful method to replace the hard coulomb potential by smooth pseudopotential, which means the core and whole electrons will be replaced by pseudo core and valence electrons in order to simplify calculation problems.
When the cut-off parameter $r_{c}$ approaches radius of atom $R$, the transferability of the obtained pseudopotential will become better. However, when $r_{c}$ is close to $R$, the wiggle of due to the orthogonality of core wave-function will reduce the accuracy and efficiency of calculation. So the balance between the smoothness and transferability of pseudopotentials should be taken into consideration.
Extended reports
- Blog note by Hongjin: Norm-Conservation Pseudopotentials(NCPP)